

Marine scientists show impact of acidification on mussel larvae
Shells have a hard, calcareous shell to protect themselves against environmental hazards and enemies. Increased acidification in the ocean makes it increasingly difficult for organisms to form their shells. Researchers from the Christian-Albrechts-Universität zu Kiel (CAU) and GEOMAR Helmholtz Center for Ocean Research Kiel have recently published a paper in the journal Nature Communications describing their discovery that mussel larvae are sensitive to ocean acidification, which leads to reduced calcification rates and shell dissolution.
Mussels are a popular seafood item. The brown-black shells occur in tidal regions of the coastal zones. However, like many marine animals which protect themselves with a calcareous shell, their shells are weakened by, among other things, the increasing acidification of seawater. This acidification is due to the absorption of additional carbon dioxide from the atmosphere, which is dissolved in seawater. The mussel is very sensitive to a decline in pH levels in its early life stages. This is mainly because of the high rate of calcification during the larval stage: between the first and second day of their lives, they form calcareous shells, which correspond to the weight of the rest of the body.
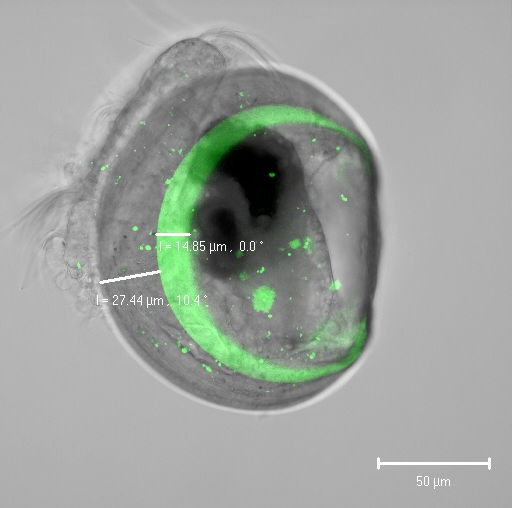
"For the first time, we used two different methods to understand the calcification of one to two-day-old shelled larvae to estimate their sensitivity to climate change," said Kirti Ramesh, first author of the study. "With the help of fluorescent dyes and specialized microscopy techniques, we were able to track the deposition of calcium carbonate in living larvae and show that calcium carbonate is not formed intracellularly, as previously thought. It is more likely that calcium is acquired directly from the seawater and transported to the shell via specific transport proteins. Then, very close to the shell, the formation of calcium carbonate takes place, “ he explained.
In the second step, the team studied the abiotic conditions directly under the shell. Calcium, pH level and carbonate were measured in minute troughs with tiny microelectrodes made of glass. "For the first time, we have been able to show that the mussel larvae are able to increase the pH and the carbonate concentration below the shell, which then leads to higher rates of calcification," said Dr Frank Melzner, from GEOMAR. "However, with increasing acidification, the pH values below the shell also decrease, which leads to reduced calcification rates and, at very high CO2 concentrations, shell dissolution and increased mortality occurs," he continued. It is interesting, however, that the shells dissolve only at very low pH values. This suggests that organic constituents of the mussel shell contribute to dissolution resistance.
"With these results, we can establish a direct relationship between the calcification rate of mussels and the carbonate chemistry of seawater," explained Prof Dr med. Markus Bleich, Head of the Physiological Institute at Kiel University. The reason for the larvae’s high sensitivity to acidification is its limited ion regulation systems, he continued.
What's next? “We will use genetic and proteomic methods to investigate which proteins play a role in the transport of calcium and carbonate, and which organic substances in the shell increase their dissolution resistance. Findings from our laboratory show that some mussel populations, especially from the Baltic Sea, are more tolerant to ocean acidification. We think that the key to increased resistance of mussel shells to dissolution lies in the variation of organic shell constituents," said Melzner. Such tolerant populations could subsequently be the winners in the climate change arena.
 Herbert
Herbert 11th January 2018
11th January 2018